In the Kobayashi Solid State Chemistry Lab.
Chemical energy conversion/storage using electrochemical devices such as fuel cells and batteries will become increasingly important for future sustainable societies. Ion conductive materials are used as electrodes or solid electrolytes, and are key for determining the performance of these devices.
Therefore, much better understanding characteristics of existing electrodes/electrolytes materials such as crystal structure, thermal stability and their reaction mechanism is key for achieving enhancement of battery performances. On the other hand, finding novel ion conduction phenomena through synthesis of a new class of substances leads to a creation of new battery systems.
Our group focuses mainly on two research topics; (i) Control of an electrode/electrolyte interface in lithium secondary batteries for enhancement of battery performance (ii) Synthesis of hydride ion (H–) conductive and development of a novel battery system utilizing the H– conduction phenomenon and H–/H2 redox reaction.
Creation of electrochemical devices based on new principles
Under construction.
Investigation into mechanism of material properties and functionalities
Neutron Scattering and Heat Capacity Measurements
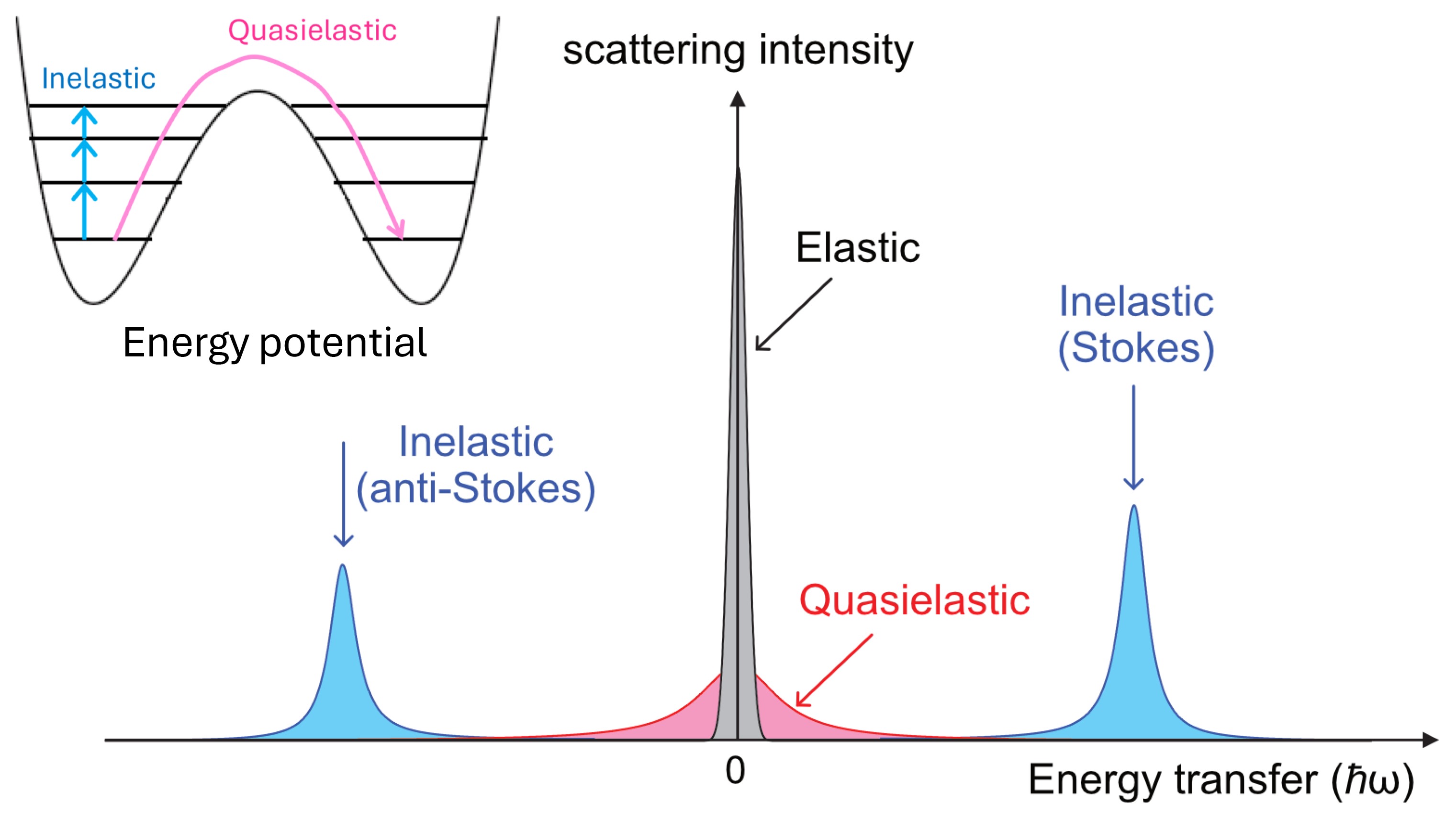
Developing high-performance ion-conductors requires not only structural analysis but also elucidating the ion conduction mechanism through physical property measurements. Our laboratory conducts inelastic neutron scattering (INS) and quasielastic neutron scattering (QENS) measurements at JRR-3 and J-PARC, alongside heat capacity measurements using a custom-made adiabatic calorimeter.
INS is a technique for observing various vibrational phenomena within materials. Unlike methods such as Raman scattering or infrared spectroscopy, INS has no selection rules and, most importantly, can provide vibrational information in the energy-momentum plane (so-called phonon dispersion relation). QENS is a method of observing jump motion and diffusion motion within materials on timescales ranging from 1 ps to 100 ns. QENS can provide spatial information of motions (e.g., the jump distance of ions) as INS. This is not possible with techniques like NMR. A common scenario is that ions undergo harmonic vibrations at specific sites within the crystal at low temperatures, then, as temperature increases, these vibrations soften, exhibiting greater anharmonicity, and eventually jump to other sites. This process can be clearly observed as a decrease in INS frequency, broadening of INS peaks, and the appearance and broadening of QENS peaks
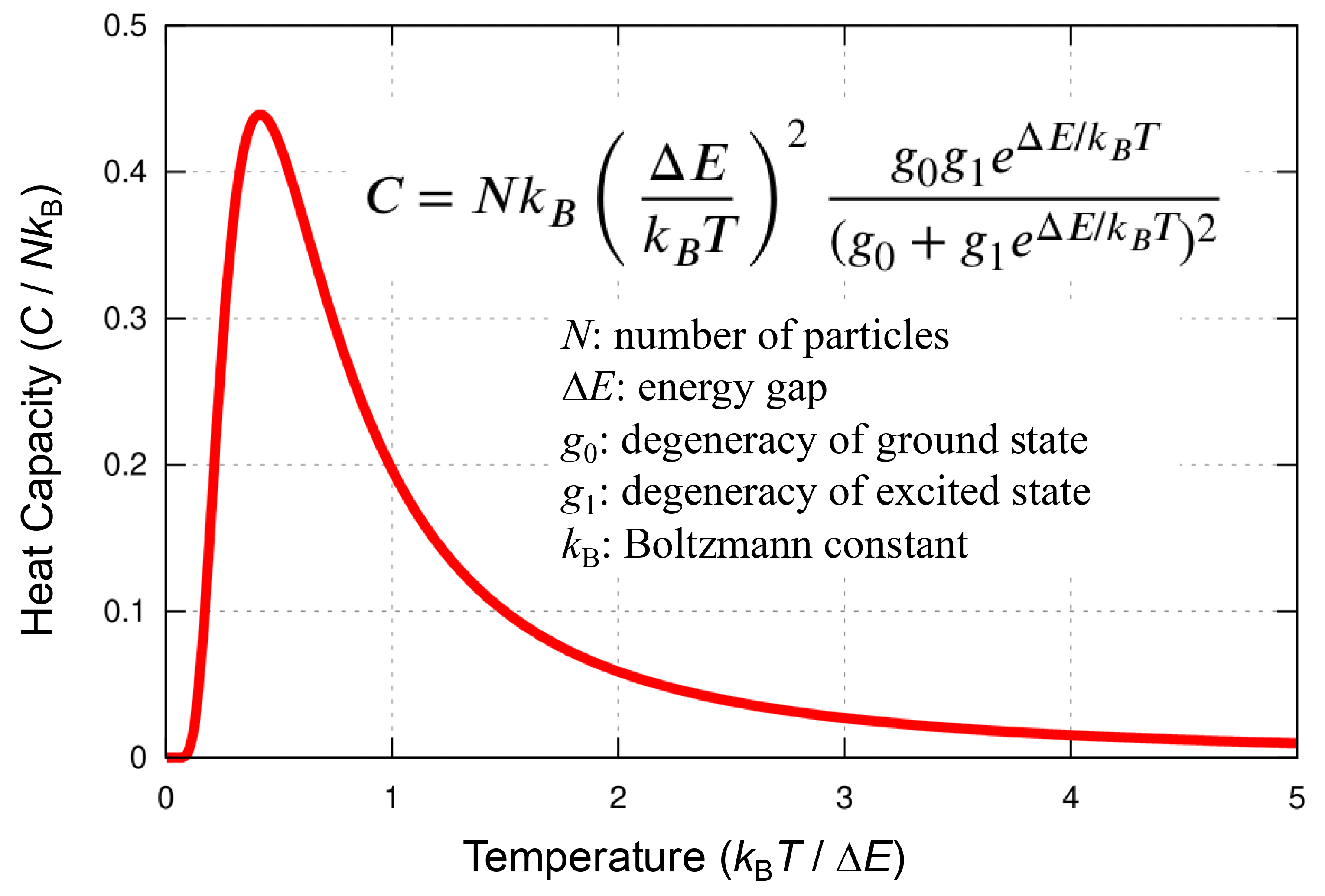
The adiabatic calorimeter offers significantly higher accuracy relative to absolute heat capacity (ΔCp/Cp < 0.1%) compared to commercially available differential scanning calorimeters (DSC) or relaxation calorimeters (PPMS), and can also observe non-equilibrium phenomena such as glass transitions. In many ionic conductors, ions, that are ordered at the most stable sites at low temperatures, become excited to metastable sites upon heating, leading to disorder and ion conduction. This process is known to be represented by Schottky heat capacity. By fitting experimental data to this model, the number of excited ions, excitation energy, and number of excitation sites can be determined.
Understanding the Physics of Ions through Thin Films
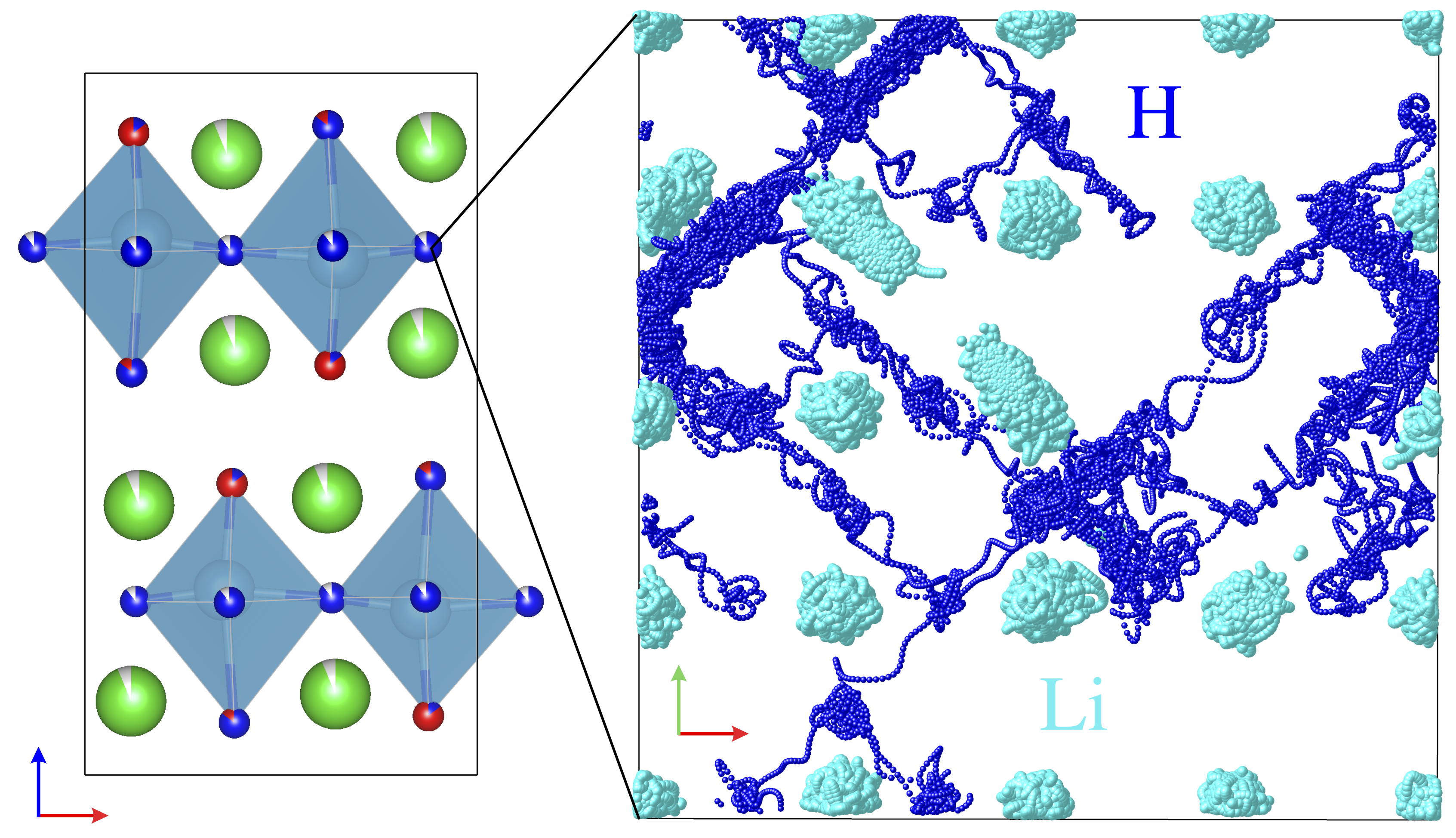
In the study of ionic materials, measurements have traditionally been carried out using powders or polycrystalline sintered samples. However, in such bulk materials, grain boundaries and defects are intricately entangled, often obscuring the intrinsic physical properties by averaging them out.
To overcome this limitation, we are pursuing research that employs thin films as an ideal platform to elucidate the fundamental nature of ionic conduction and phase transitions.
By fabricating materials in thin-film form, we can achieve:
- Control of single-crystal-like structures with well-defined crystallographic orientation
- Stabilization of novel phases induced by lattice strain and interfacial effects
- Visualization and separation of ionic transport processes at electrode interfaces
These approaches allow us to precisely analyze the intrinsic ion transport properties and structure–property relationships that cannot be observed in powder samples.
Computational Science Approach
Our laboratory focuses on theoretical investigations of materials at the atomic and electronic levels using computational approaches such as first-principles calculations and molecular dynamics simulations. In particular, we are interested in metal hydrides, which are promising candidates for safe and efficient hydrogen storage in future energy systems
A key aspect of our research is to understand the mechanisms of ion diffusion—especially hydrogen and metal ions—within these materials. By analyzing diffusion coefficients and migration pathways at the atomic scale, we aim to gain insight into the stability and reactivity of hydride materials. Since these simulations require substantial computational resources, we utilize supercomputers to perform high-accuracy calculations that are often difficult to achieve through experiments alone.
Theoretical insights obtained from our simulations contribute to the design and prediction of novel materials, supporting the development of innovative technologies in the energy and environmental fields. By combining computational materials science with fundamental physical chemistry, our lab is committed to advancing foundational research for a sustainable future.
Exploration for novel functional materials
Perovskite Compounds for Ammonia Storage
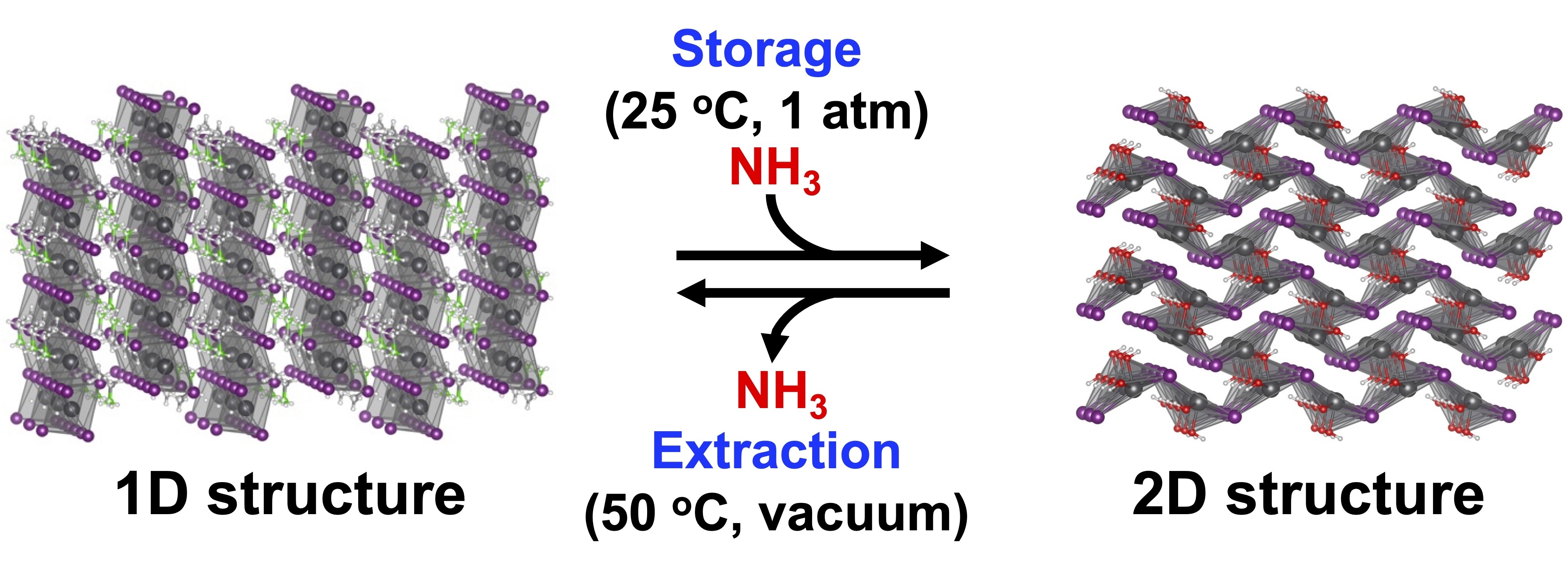
Ammonia (NH3) is expected to be utilized not only as fertilizer and a chemical raw material, but also as a hydrogen carrier and a decarbonized fuel. However, NH3 is a corrosive gas and requires liquefaction at low temperature or high pressure for storage. Therefore, a safe and easy way to store NH3 is long awaited.
We have developed organic-inorganic hybrid perovskite compounds that store NH3 at ambient temperature and pressure. The perovskite compound showing a one-dimensional (1D) structure is transformed into a two-dimensional (2D) structure by chemical reaction with NH3. NH3 undergoes chemical conversion to low-corrosive nitrogen compounds. The stored nitrogen compounds are extracted as NH3 at 50 °C under vacuum, while the perovskite compound reverts to its original 1D structure. The reversible structural transformation of the perovskite compound allows for repeated storage and extraction of NH3.
The perovskite compounds enable the easy and convenient storage of NH3, contributing to establishment of sustainable NH3 storage.
Materials Science for batteries
Under construction.

 RIKEN
RIKEN
